Abbott lands FDA Approval for heart stent
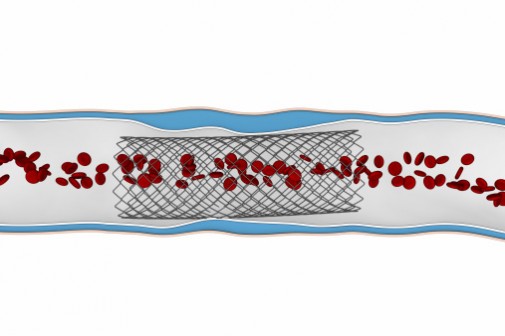
Abbott Laboratories announced that its Xience Xpedition heart stent was approved by the U. S. Food and Drug Administration and will be launched in the United States immediately.
A heart stent is a tiny tube that is placed into a diseased blood vessel to keep it open so that blood can flow properly to the heart. Abbott’s stent delivers a drug called everolimus, which helps to keep the vessel from reclogging.
Each year in the U.S., about 600,000 stents are inserted into ailing hearts, according to a report published in the Journal of the American Medical Association. Of those, about 71 percent were done during emergencies, such as heart attacks.
Advocate Health Care’s nationally ranked cardiovascular programs offer patients access to pioneering protocols and technologies like the use of heart stents and other devices in treating patients who suffer from heart disease. Last year, Advocate doctors performed more than 1,300 open heart surgeries, more than any other health system in Illinois.
Click here to learn more about heart services offered at Advocate.
Related Posts
Comments
About the Author
health enews staff is a group of experienced writers from our Advocate Health Care and Aurora Health Care sites, which also includes freelance or intern writers.

















